
Akshar Global Clinical Research Pvt Ltd. is a leading provider of comprehensive clinical research services to global pharmaceutical, biotechnology, and medical device companies. Specializing in efficient trial management across diverse therapeutic areas including oncology, cardiology, neurology, and infectious diseases, etc. we offer expert guidance from protocol design to regulatory approval. Our services encompass end-to-end services of Clinical trials, robust data management, biostatistics, and meticulous medical writing to ensure accurate and timely insights. With a commitment to quality and ethical conduct, we leverage cutting-edge technologies to streamline processes and deliver actionable results. Operating on a global scale, we facilitate broad patient recruitment and comprehensive data collection through strategic partnerships worldwide. At Akshar Global Clinical Research, our mission is to accelerate the development of innovative healthcare solutions, adhering to the highest standards of integrity and regulatory compliance.

ClinGroup is a global health sector solutions provider. Established in France, we offer innovative solutions to all health sectors needs in international standards quality/accreditation, digitalization, products registration, regulatory and maintenance, and throughout their full R and D programs. We are the first philanthropic international comprehensive sustainable health solutions provider.

We translate science into treatment, with patients for patients – rethinking clinical trials.
CTC was founded in 2011 and has, since then, participated in over 550 industry sponsored clinical trials on behalf of small start-ups as well as large international pharmaceutical companies and CROs.
Our track record includes, but is not limited to, first-in-human (FIH), PET/MR, early precision QT (EPQT), drug-drug and food interaction, pharmaco-EEG, nicotine, biosimilar, bioequivalence, and bioavailability studies, new drug delivery systems, medical device investigations and Phase II studies. We also participate in international, multicentre Phase III-IV studies in patients with various diagnoses.
Our Site Network work in close collaboration with the Clinical Operations, Biometrics, Pharmacovigilance, DMPK & Clinical Pharmacology, Bioanalysis and Formulation Development departments to provide a true full-service option to our customers. The Site Network consists of six state-of-the-art clinics across Sweden, and one in the Netherlands.
Our service offering is: Full service CRO capabilities within early-phase clinical trials (Phase 0-II) including project management, medical writing, monitoring, pharmacovigilance (pre- and post-marketing), biometrics, DMPK, bioanalysis and formulation.

OPIS, founded in 1998 by medical doctors from the pharmaceutical industry, is a global full-service CRO with 26 years of experience across diverse therapeutic areas. We provide 360° clinical trial support—from study concept and protocol development to full project execution, data management, and final reporting. OPIS manages international Phase I–IV interventional, non-interventional, and medical device studies, including pre- and post-marketing investigations for diagnostics, nutraceuticals, and food supplements. With over 1,500 studies managed, our specialised teams deliver high-quality results through scientific, medical, and statistical expertise. OPIS combines a global reach with deep local regulatory knowledge to accelerate the development of safe and effective medical therapeutics.

OSMUNDA, a global leading technology service provider for medical devices and in-vitro diagnostics (IVDs) has grown to stand as a frontrunner in providing comprehensive consulting services, clinical trial and global registration services. As a third-party service provider for medical devices and IVDs, the firm offers one-stop medical device industry solutions for developers, manufacturers, users and governments worldwide.

EBS CRO is a full-service Contract Research Organization based in Serbia, with over 20 years of experience supporting pharmaceutical and biotech companies in clinical development. We offer comprehensive services across all phases of clinical trials, including regulatory support, project management, site monitoring, and more.
Thanks to our in-depth knowledge of local regulations, strong relationships with investigators and sites, and a flexible, hands-on approach, we ensure trials are conducted efficiently and with high quality. Our team is known not only for smooth study execution but also for stepping in when trials need support — bringing proven experience in study rescue through fast start-up, responsive sites, and readily accessible patient populations.
Whether you’re running early-phase studies or complex multicenter trials, EBS CRO can help you move forward — on time, on budget, and with confidence.

Elevate CRO is a full-service contract research organization partnering with pharmaceutical companies and CROs worldwide. Our team has the expertise to navigate Turkey’s regulatory environment efficiently, managing approvals with ethics committees and the Ministry of Health.
We support a broad range of therapeutic areas, including hematology, oncology, rare diseases, and infectious diseases. Operating under a strong Quality Management System with ISO 9001 certification, we maintain strict compliance and high standards at every step.
Regular internal audits reinforce quality by ensuring adherence to procedures and identifying areas for improvement. Continuous training is central to our culture, fostering professional growth while strengthening our commitment to excellence.
With regulatory expertise, robust quality oversight, and strong professional relationships, we ensure clinical trials progress smoothly and without unnecessary delays, delivering the reliability our partners expect.

Boji Medical Technology Co., Ltd is an end-to-end global CRO headquartered in China.
Since its founding in 2002, Boji Med has evolved to provide full clinical and regulatory services to include regulatory affairs, preclinical research, clinical trials management for phase I~IV, PMS, clinical trial for medical devices, data management, and biostatistics.

BIOTrialMed is a specialized clinical research organization (CRO) focused on advancing biomedical innovation through expert clinical trial execution. The company’s core business spans the full spectrum of clinical development, including study design, protocol development, regulatory strategy, and trial management across all phases — from Phase I to Phase IV. BIOTrialMed provides end-to-end services such as site selection, patient recruitment, clinical monitoring, data management, biostatistics, and medical writing. Backed by a team of experienced professionals, the company supports biotech, pharmaceutical, and medical device clients in navigating complex regulatory environments and accelerating the development of safe and effective therapies. With a commitment to scientific rigor, operational excellence, and patient-centric solutions, BIOTrialMed plays a vital role in bringing innovative healthcare solutions to market.

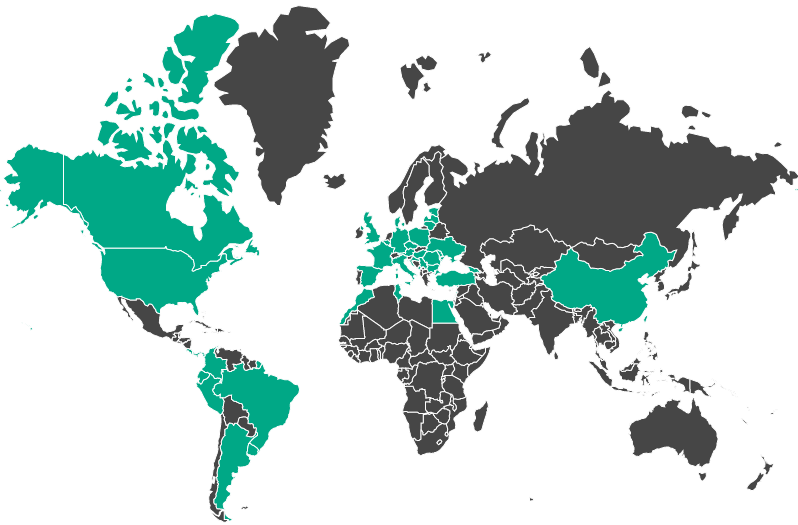
Find a partner in a certain geographic region using