CRS is a specialty CRO and executive management consulting firm that fits-for-purpose veteran life sciences executives, attorneys, regulatory scientists and strategists, clinical operations, project management, quality assurance engineers, and sales and marketing teams to take on the biggest challenges for start-up, mid-sized, and large life sciences companies, many of whom are developing novel, first-in-class drugs, biologics, diagnostics and medical devices, or software as a medical device (SaMD). Service areas are clinical trials, Functional Service Provider (FSP) models, selection and management of CROs, regulatory compliance strategy and submissions, QMS, EU MDR and GDPR, rescue trials, TMF, risk mitigation and remediation, training high-performing teams, staffing solutions, and decentralized trials.
Indications represented are precision medicine, immuno-oncology, central nervous system, infectious diseases, respiratory diseases, orthopedics, de novo devices, digital health and mobile apps, ophthalmology, reproductive health, regenerative medicine, cardiology, major depressive disorder, opioid-sparing pain, cannabis, radiology, and critical care. CRS is a qualified service provider for US federal and military grants.

Established in 2002, GCP is today a leading Israel based, full-service Contract Research Organization (CRO) and Clinical Research Training Center. For two decades we have delivered successful Clinical Trial Management, Biostatistics and Data Management solutions across a wide range of therapeutic areas for pharmaceuticals and medical device companies. Our services support the entire research process from protocol development, site feasibility and selection, site monitoring and management to final study report.
The courses delivered by our Clinical Research Training Center cover all aspects of managing clinical trials.

At CroxxMed, we are a team of experienced personnel striving to make your experience as smooth as possible. Nordic by nature, we provide complex knowledge built on a solid foundation of years in clinical research within many different therapeutic areas. CroxxMed offers services in all phases (I-IV); clinical operation, regulatory affairs, data management, statistics, medical writing, pharmacovigilance, and consulting services for all types of medical device studies. We are at all times updated on the market. At any time, fully in tune with the regulatory requirements and trends in the industry. We are less boring than average, and you are our top priority.
We have a flat infrastructure, short communication routes, efficient dialog, and only experienced employees.

Research Professionals (RP) is a clinical CRO offering a wide range of clinical research services to provide clients with access to excellent study sites and experience in 15+ therapy areas. RP staff has broad experience in CRO services for phase I.-IV. biopharmaceutical trials, medical device trials and home care clinical studies. RP’s dedicated team gained strong industry expertise working at large pharmaceutical, biotech and CRO companies. RP’s leadership believe that the new way of serving market needs increases efficiency that leads to a victory for all participants – from sponsor to patient. RP bounds up with 150 associated staff – including research nurses. RP is fully dedicated to the highest standards of conducting clinical trials by CenterWatch SOPs and its trial activities are covered by HDI Insurance Services. The combination of local knowledge, global experience, and the latest technology allows our clients to conduct their studies faster and more cost-effective.

International Drug Development Institute (IDDI) is a therapy-focused expert organization in biostatistical, clinical data management and integrated eClinical services that is committed to assisting pharmaceutical, biotech, medical-device and nutrition companies, as well as academic institutions, in several disease areas, including oncology and ophthalmology. IDDI optimises the clinical development of drugs, biologics and devices thanks to proven statistical expertise and operational excellence.
Our services include Study Design, randomization and trial supply management (RTSM), Electronic Data Capture (EDC), Clinical Data Management, Biostatistics, Independent Data Monitoring Committees (IDMC), Biomarker validation and Personalized medicine.
Our experience amounts to 1200+ clinical trials (Phase I – IV), 850+ Scientific publication, 750+ scientific conferences and 350 happy customers. Science is part of our DNA.
Founded in 1991, IDDI is privately-owned, counts 150 employees and has offices in Belgium, and Raleigh (NC).

CC Clinical research Consultants is a clinical and regulatory affairs CRO (Contract Research Organization), based in central Paris, that provides clinical trials support to the Medical Industries throughout Europe. Assignments include regulatory and ethical initiation, full study management and monitoring, safety reporting, per European and local country requirements. Monitors assigned to a project are experienced, local country language speakers, and are located in or close to the assignment locations.
Industry experience of the upper management allows adequate understanding of clinial development needs, startup specificities, medical device particulars, regulatory requirements, teamwork and network building needs, deadline settings as well as budget implications and constraints.
Flexibility is a key feature of this medium-sized CRO, enabling adaptation to specific needs through direct interaction between the management of the CRO and the Sponsors and KOLs.

Anagram does clinical research since 1998, with Investigational Products and Medical Devices, from first-in-human to post-registry studies. With experience in most therapeutic areas we are highly specialized in Cardiology, CNS (stroke) and Oncology. Our 20 years’ experience with medical device (MD) clinical trials, have positioned Anagram as a leader in this area (MD and diagnostic kits).
Anagram and IEC merged in 2018 and provides integrated full service CRO + Imaging Core lab services relating to:
Clinical Monitoring
Project Management
Regulatory
Consulting
DM & biostatistics
Medical Imaging core lab services (IEC)
We have extensive experience working with small / medium sized clients, who need advice and close support to design, plan and run their clinical research projects. While we work at international level, our main expertise is in Spain, where we provide high quality data with a cost effective and efficient approach. Our imaging Core Lab provides central reading and imaging analysis pertaining to patients in clinical trials, providing high quality data which can be directly exported to the eCRF.

Over 30 years of experience in managing medical device clinical investigations around the globe.
Regulatory and quality experts to get medical devices registered rapidly and efficiently. Regulatory strategy development
Because medical device clinical investigations are highly specific, our team of experienced, (para)medically and multilingual monitors and project managers are exclusively dedicated to medical devices and work according to our certified ISO 9001 quality system while taking into account cultural nuances to save you critical time and resources for clinical investigation management throughout Asia, Europe, South America and the Middle East.
Our services are Global clinical strategy development including reimbursement, Clinical Evaluation Report writing, Protocol design and development including statistics, Investigator brochure writing, Instructions for use, CRF development, Site selection, Clinical trial agreements, Regulatory and Ethics Committee submissions, Clinical project management, Product training, Site monitoring, Medical monitoring – Safety reporting, Clinical report writing, Data management and biostatistics, Clinical auditing.

MEDEX is an organization that specializes in managing clinical research sites. We provide services for Contract Research Organizations (CRO’s), pharmaceutical companies, biotechnology companies, and medical device companies.
Clinical trials frequently fail to reach its aim by reason of insufficient enrollment, poor compliance and performance. Proactive approach to site management is therefore a cornerstone of a clinical trial.
Site Management Services from MEDEX take the burden off investigators so that they can focus on the patient, and ensure that the study is conducted at the highest standards of quality, ethics and performance.

KLIFO is an integrated North European drug development consultancy with significant experience in partnering with pharmaceutical and biotech companies.
At KLIFO, we offer end to end solutions across all drug development areas, including strategic project management, regulatory affairs, clinical development, clinical trial supply, quality assurance (QA), Chemistry Manufacturing and Controls (CMC) development, non-clinical development and pharmacovigilance concerning the development of pharmaceutical products and medical devices.
KLIFO Clinical Operations Solutions has a strong track record of conducting early and late stage clinical studies and investigations in an efficient and flexible manner. During the last three years, KLIFO has supported more than 60 clinical studies and clinical investigations globally within various therapeutic areas, including oncology, gastroenterology and CNS, as well as different device classes.
KLIFO Clinical Operations Solutions also offers consultancy services and solutions for specific tasks, competence- and resource gaps on short- or long-term basis.

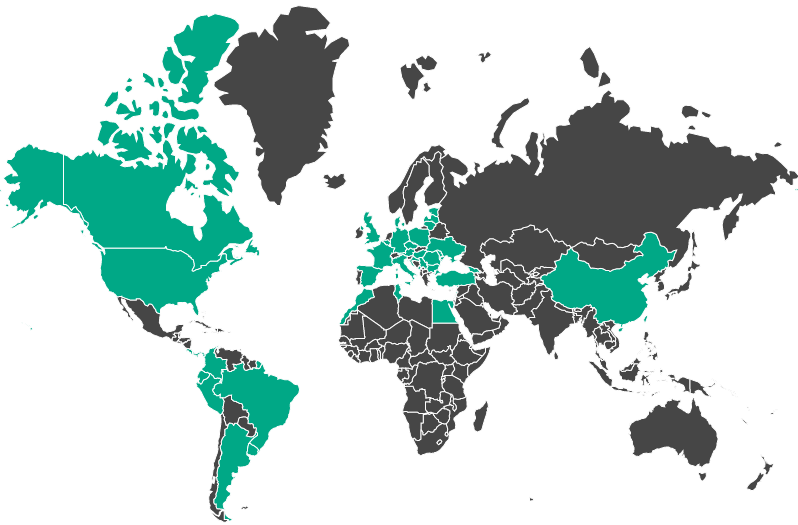
Find a partner in a certain geographic region using