Aixial is a mid size, full-service CRO with global reach. Over the past 25 years, Aixial has worked in collaboration with small biotechs, multiple mid-size pharmaceutical companies, and five of
the top ten pharmaceutical companies in over 1,400 studies.
We are a specialized clinical development partner, committed to deliver Phase I-IV clinical trials, as well as pre-clinical and post-market support.
With a focus on developing long term partnerships, Aixial is recognized as a preferred service provider for many companies. We
have a track-record for successful execution of clinical trials with an approach to project delivery that is known to enable timelines to be met consistently.

As a local contract research organization, we conduct multicenter and single center clinical studies within Bulgaria.
Our capabilities cover A – Z clinical research services starting from: feasibility assessment, study teams and sites selection, budget management, contracts negotiation, medical writing, regulatory activities, investigator meeting organization, study start-up support, study team training, project management, subjects recruitment, sites management, vendors management, clinical monitoring, safety reporting, auditing, data management, biostatistics, final report development and scientific consulting.
On the other hand, our privately-owned center for early phase human and drug research with a built-in phase I unit enables us to conduct a variety of single-center clinical research on healthy volunteers and specific patient populations:

Marti Farm Ltd., company based in Zagreb, Croatia is a contract research organization supporting the needs of the companies working within pharmaceuticals, biotechnology, medical devices, food supplements, cosmetics and healthcare industries across a variety of consulting and operational services.
Mart Farm Ltd.provides the following services:
We are internationally recognized as a reliable partner and provider of consultancy and regulatory services with a proven track record of 9+ years. Our core belief is continuous personal development aimed at strengthening our expertise and approaches in order to drive added value for our clients from both the operational and the strategic aspects.
Over the last several years, we have been increasingly expanding our business internationally in search for new satisfied clients and trusted partners.

We are a Contract Research Organization (CRO) approved by the Drug Regulatory Authority of Pakistan (DRAP) – Ministry of National Health Services, Regulations, and Coordination (NHSRC). Covance Pharma Solution is committed to providing our partners with smart and innovative solutions to ensure that their requirements are met in the most cost-effective, high-quality, and timely manner possible. We achieve this through collaborative and synergistic partnerships founded on the principles of quality, transparency, and integrity. Covance Pharma Solutions provides a comprehensive range of Clinical trial and Bioequivalence services in Pakistan based on CORE values of Compliance, Optimism, Regulation, and Ethics.

Being a leading contract research organization (CRO) in Taiwan, Bestat is dedicated to offer clinical services with the highest quality.
As experienced professionals within the industry, we provide customized services, which can be deployed on a stand-alone basis or as part of an integrated “comprehensive-service” solution, including:
Our teams always have the good professional spirit with highly efficiency and cooperated attitude. You can trust that we will be your good partner. So we are the best choice for you. Welcome to contact us if you are interested in Bestat.

Brillance is independent, Polish CRO specialized in monitoring and management of phase I-IV clinical trials (medical products and devices) in Poland, Czech Republic and Slovakia. Since 2004 offers services both for local and local parts of global projects for pharmaceutical, biotech or other CRO companies. Regionally operating offers a deep insight into local markets thanks to excellent relationships with sites, staff and knowledge of local authorities requirements. Brillance deals with trials in almost all therapeutic areas, most currently in (hemato)oncology and cardiology. It is well known for flexibility, patients quick recruitment and delivering quality data. Professional regulatory team handles challenging cases and its lawyers cover demanding sites/investigators contracting. US Clients with no presence in EU are guided and represented by Brillance locally.

SMO-Pharmina is providing superior clinical trial management services in Georgia: ensuring quality care for patients, adherence to international guidelines and delivery of reliable data.
Since 2006, LTD SMO-Pharmina has contributed to the establishment of GCP compliant clinics. Nowadays, the company has set up the renewable database of clinical trial sites and Principal Investigators in all therapeutic areas. SMO-Pharmina has worked with up to 100 clients as SMO or CRO. Over 100 sites managed by SMO-Pharmina have undergone about 140 successful audits and 2 FDA inspections.
SMO-Pharmina ensures effective management of the entire project from start-up to close-out and offers the clients the following services: Site Selection, Project Management, Regulatory Affairs, Site Monitoring, Study Coordinator support, Training Center, Clinical Laboratory, Pharmaceutical Warehouse, Archive, Distribution of clinical trial materials, Logistic support.

AnRes Clinical is a small CRO/consultancy, composed of a small team of passionate clinical Project Managers, Clinical Research Associates, Regulatory Submission specialists, Site Contract Managers, Medical Consultants, Statisticians, PV Experts and Regulatory Affairs professionals with a wealth of experience. We mainly work on a freelance basis. In addition to that we partnered with a clinical database development partner who can create eCRFs, eQuestionnaires, ePatient Daries, ePROs and many other databases tailored to study needs. Our current global reach includes Scandinavia, Europe and United States. What makes us different is passion, wealth of experience and collaborative approach. We specialise in running clinical trials in Rare diseases, Oncology, Neurology and Cardiology medical devices, however we also work in a number of other therapeutic indications. Our mission is to partner with biopharmaceutical companies and Clinical Research Organisations in order to support development of better medicines.

BlueClinical is a privately-owned CRO, founded in May of 2012 with the aim of providing scientific consultancy and performing research & development activities for medicines, medical devices, diagnostics and other healthcare products.
BlueClinical is organized in 3 business units:
– BlueClinical Phase I: devoted to the conduct of bioequivalence and Phase I clinical trials in healthy subjects and selected patient population, which are carried out in our human pharmacology unit, located at Hospital da Prelada (Porto, Portugal).
– BlueClinical R&D: acts as a full-service CRO for clinical trials in patients in Portugal and Spain, and provides consultancy and standalone services in medical writing, clinical monitoring, pharmacometrics, regulatory affairs, pharmacovigilance, clinical data management, clinical data programming, biostatistics, project management.
– BlueClinical CRP (from “Clinical Research Partnership”): provides in loco clinical research site management services to 9 institutions belonging to the National Healthcare System. With the support of the other business units, it operates two Early Phase Clinical Trials Units (EPCTU) dedicated to Phase I studies in special populations (e.g., hepatic impairment, renal impairment) and Phase IIa studies (including proof of concept, POC) in target therapeutic indications.

Aesculape CRO is a small CRO with presence in Europe and Asia. We have offices in Belgium, Spain, Italy, Poland, Hungary, Singapore, Malaysia, India, Thailand, Vietnam, Indonesia and the Philippines. We are offering full services (with subcontracting DM activities) in most therapeutics domains (Oncology, CNS, Cardio metabolic, ID, NCE, Biosimilars, medical devices, nutraceuticals)
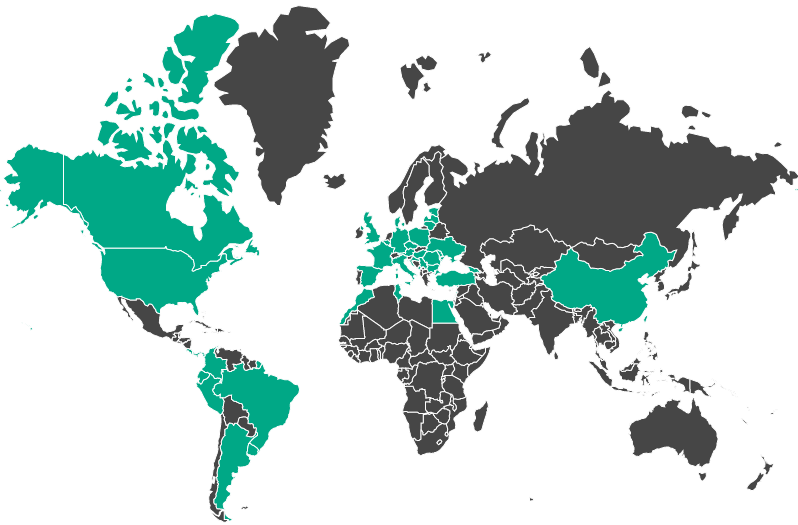
Find a partner in a certain geographic region using